Regulation Monitoring
Why do I need to keep track of new and updated rules?
Product regulations can be amended, which results in new and updated requirements (i.e., labelling requirements). Further, new regulations are added each year which can also impact your product.
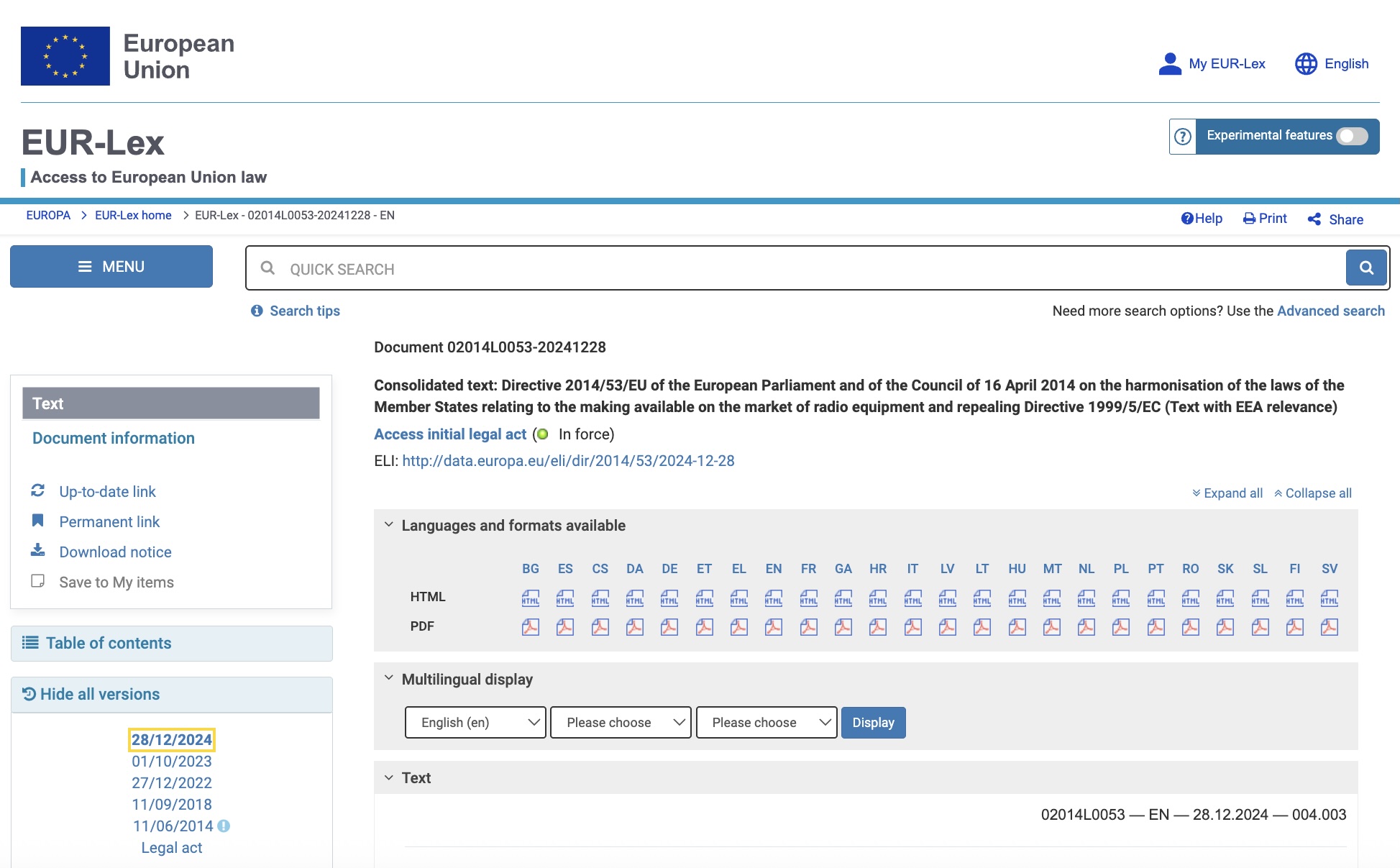
Example: EUR Lex (we also track US, UK, Canadian, and Australian sources)
Each month, we check hundreds of regulations in EUR Lex, eCFR, Legislation.gov.uk, Laws-lois.justice.gc.ca, and Legislation.gov.au. Our goal is to find out if monitored regulations have been updated.
This is critical, as regulatory updates can result in new or changed requirements directly impacting products.
Note: We also update the Compliance Gate Platform if we detect updated regulations, directives or other legislation. That said, note that not all entries are actively monitored.
Example: EU Official Journal (we also track US and UK sources)
We also stay on the lookout for new product regulations. To do so, we monitor several official sources in the US, EU, UK, Canada, and Australia.
Note: We frequently add new regulations to the Compliance Gate Platform, in part based on our monitoring activities.
Each month, you’ll receive a report listing new and updated regulations
Sources / Monitoring / Update Methodology
Please read the US / EU / UK / CA / AU Database documents for details on monitored sources, and how we manage updates in the following documents.
You can find a list of limitations and risks related to source monitoring in this document. 1. We monitor certain legislation texts and other sources to keep track of new versions of regulatory requirements covered by the CRL database. 2. You can learn more about monitored sources and our methodology here: 3. We send a report each month summarising our findings and specifying if we deem it necessary to update CRL entries. 4. We also monitor news and announcements concerning new regulations and compliance requirements (not already covered by the CRL database). You can find a list of limitations and risks related to source monitoring in this document. We review sources once per month. For example, we review updates from April 2026 in May 2026. Please open the US / EU / UK / CA / AU Database documents to learn more about platform updates. No, we cannot guarantee that the Compliance Requirements List database is always up to date as we do not implement changes in real-time. Each month, we review updates from the previous month. We also assess if an update to the affected CRL entry is necessary. As such, CRL entries are only updated after sources have been updated. It is therefore critical that you always take action based on the latest version of the relevant legislation, guidance documents, and other sources. Further, document templates are only updated during the annual audit. 1. We normally send a Monthly Report on the 2nd or 3rd Tuesday of each month. 2. You can also find a list of updates in the Compliance Gate Platform update log. The Monthly Report is a compilation of compliance news and updates from monitored sources in the US, EU, UK, Canada, and Australia. It also helps us to decide internally whether we need to update the Compliance Gate Platform. The report can help you stay up to date with new and updated compliance requirements, including those that do not result in an update of the Compliance Gate Platform. You will receive a report each month, for the duration of your subscription.Feature Overview
What sources do you monitor?
Limitations and risks
How often do you review the sources?
How do you keep the platform up to date?
Do you guarantee that the Compliance Gate Platform is always up to date?
How are we made aware of news and updates?
What is the purpose of the Monthly Report?
How does the report benefit us?
How many reports do we receive each year?