Request a free EU Authorised Representative Quotation Here
The Market Surveillance Regulation (EU) 2019/1020 states that sellers based outside the EU selling certain products directly to consumers in the European Union must have an authorised representative. This authorised representative must based in the EU, and manage certain tasks within the market.
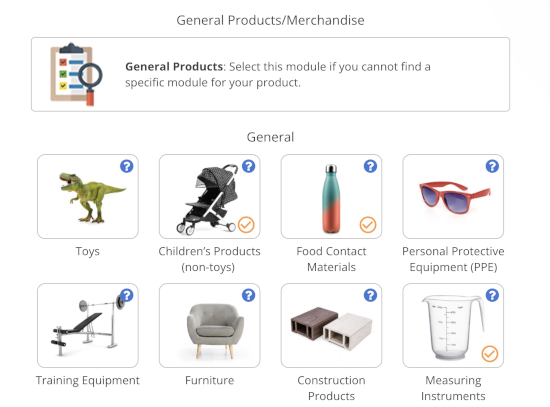
Previously, this primarily affected companies selling electronics, toys, PPE, and other products requiring CE marking. However, the General Product Safety Regulation (GPSR) extends these requirements to virtually all consumer products sold to consumers in the EU by companies established outside the EU.
In this guide, we list companies offering EU authorised representative services for products covered by the Market Surveillance Regulation and the General Product Safety Regulation.
Learn more
a. European Union Authorised Representatives: A Complete Guide
b. Authorised Representatives under the General Product Safety Regulation
Continue reading List of European Authorised Representative Companies 2025










 Request a free UK Authorised Representative Quotation Here
Request a free UK Authorised Representative Quotation Here
 Create compliance checklists for your product (US, EU & UK)
Create compliance checklists for your product (US, EU & UK) 20+ product certificate templates
20+ product certificate templates Create label files
Create label files Book product testing
Book product testing